|







|
|
Bioactivity associated with
fumonisins
Fumonisins have been associated
with the development of liver and kidney cancer in humans (Howard et al.,
2001). Fumonisins typically only exerts low levels of acute toxicity,
however horses and pigs are more susceptible than other production animals
and ingestion can cause equine leukoencephalomalacia (liquirifaction of the
brain matter in horses) and porcine pulmonary oedema syndrome in pigs (Marasas
et al., 1988). Diseases that might be a result of Fumonisin B1’s inhibitory
effects on ceramide synthesis (Wang et al., 1991).
The fumonisin biosynthesis has been described in G. fujikuroi (anamorph
F. moniliforme), but the biosynthetic pathway in F. graminearum
is believed to be similar. Elucidation of the genetic basis for
fumonisin biosynthesis was initiated with a classical linkage analysis in
1996, followed by studies in 1999 and 2001 which identified a cluster of
five genes required for synthesis (Desjardins et al., 1996;Proctor et al.,
1999;Seo et al., 2001). Subsequent mapping and characterization of the
surrounding genes resulted in the identification of 16 co-expressed genes
(Figure 13), responsible for a highly complex biosynthetic pathway, which
included a iPKS, two fatty acid synthases and numerous different classes of
modifying enzymes, including monooxygeases, dehydrogenases, an
aminotransferase and a dioxygenase (Proctor et al., 2003).
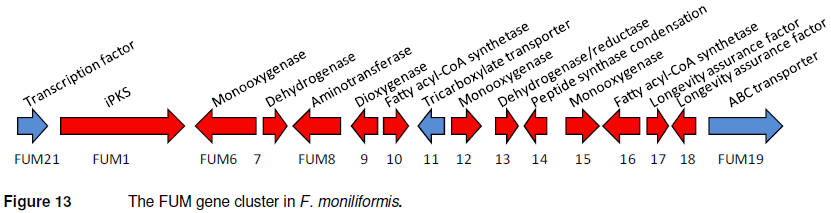
Identification of this gene
cluster allowed for the formulation of a biosynthetic model (Figure 14)
which also offered an explanation for the different types of reported
fumonisins (B1, B2, B3 and B4). Presently only FUM1 has been linked to the
formation of the polyketide backbone and FUM9 to hydroxylation of the C5
position, meaning the the majority of the suggested steps remain to be
experimentally proven (Butchko et al., 2003).
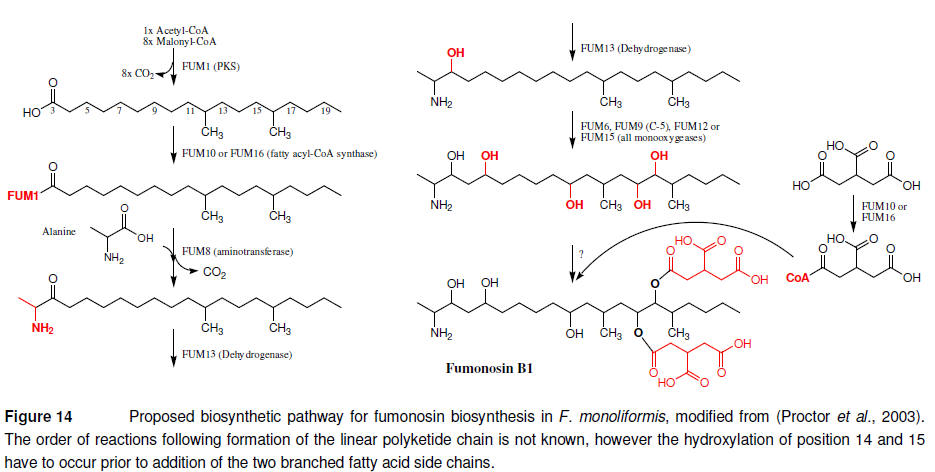
The involved PKS (FUM1) in
addition to the core domains also include KR, DH, ER and CMeT domains.
Fumonisins are nitrogen containing polyketides and could in theory as is the
case of fusarin C, be expected to include NRPS related domains. However,
here the amino group must be added by an external factor such as FUM8, an
aminotransfearase, also encoded by the gene cluster (Brown et al., 2007).
Further analysis of the FUM gene cluster has resulted in the identification
of a cluster specific transcription factor (FUM21), located adjacent to the
iPKS. FUM21 belongs to the binuclear zinc cluster class of transcription
factors, and is the prime candidate for a FUM gene cluster specific
transcription factor (Brown et al., 2007).
References
 |
Howard,P.C., Eppley,R.M., Stack,M.E., Warbritton,A.,
Voss,K.A., Lorentzen,R.J. et al. (2001) Fumonisin B-1 carcinogenicity in a
two-year feeding study using F344 rats and B6C3F(1) mice. Environmental
Health Perspectives 109: 277-282. |
 |
Marasas,W.F.O., Kellerman,T.S., Gelderblom,W.C.A.,
Coetzer,J.A.W., Thiel,P.G., and Vanderlugt,J.J. (1988)
Leukoencephalomalacia in A Horse Induced by Fumonisin-B1 Isolated from
Fusarium-Moniliforme. Onderstepoort Journal of Veterinary Research 55:
197-203. |
 |
Wang,E., Norred,W.P., Bacon,C.W., Riley,R.T., and
Merrill,A.H. (1991) Inhibition of Sphingolipid Biosynthesis by Fumonisins
- Implications for Diseases Associated with Fusarium-Moniliforme.
Journal of Biological Chemistry 266: 14486-14490. |
 |
Desjardins,A.E., Plattner,R.D., and Proctor,R.H. (1996)
Linkage among genes responsible for fumonisin biosynthesis in
Gibberella fujikuroi mating population A. Applied and Environmental
Microbiology 62: 2571-2576. |
 |
Proctor,R.H., Brown,D.W., Plattner,R.D., and
Desjardins,A.E. (2003) Co-expression of 15 contiguous genes delineates a
fumonisin biosynthetic gene cluster in Gibberella moniliformis.
Fungal Genetics and Biology 38: 237-249. |
 |
Proctor,R.H., Desjardins,A.E., Plattner,R.D., and
Hohn,T.M. (1999) A polyketide synthase gene required for biosynthesis of
fumonisin mycotoxins in Gibberella fujikuroi slating population A.
Fungal Genetics and Biology 27: 100-112. |
 |
Seo,J.A., Proctor,R.H., and Plattner,R.D. (2001)
Characterization of four clustered and coregulated genes associated with
fumonisin biosynthesis in Fusarium verticillioides. Fungal Genetics
and Biology 34: 155-165. |
 |
Butchko,R.A.E., Plattner,R.D., and Proctor,R.H. (2003)
FUM9 is required for C-5 hydroxylation of fumonisins and complements the
meitotically defined Fum3 locus in Gibberella moniliformis. Applied
and Environmental Microbiology 69: 6935-6937. |
 |
Brown,D.W., Butchko,R.A.E., Busman,M., and Proctor,R.H.
(2007) The Fusarium verticillioides FUM gene cluster encodes a
Zn(II)2Cys6 protein that affects FUM gene expression and fumonisin
production. Eukaryotic Cell 6: 1210-1218. |
|
